- Home
- Industries
- Healthcare
Loading...
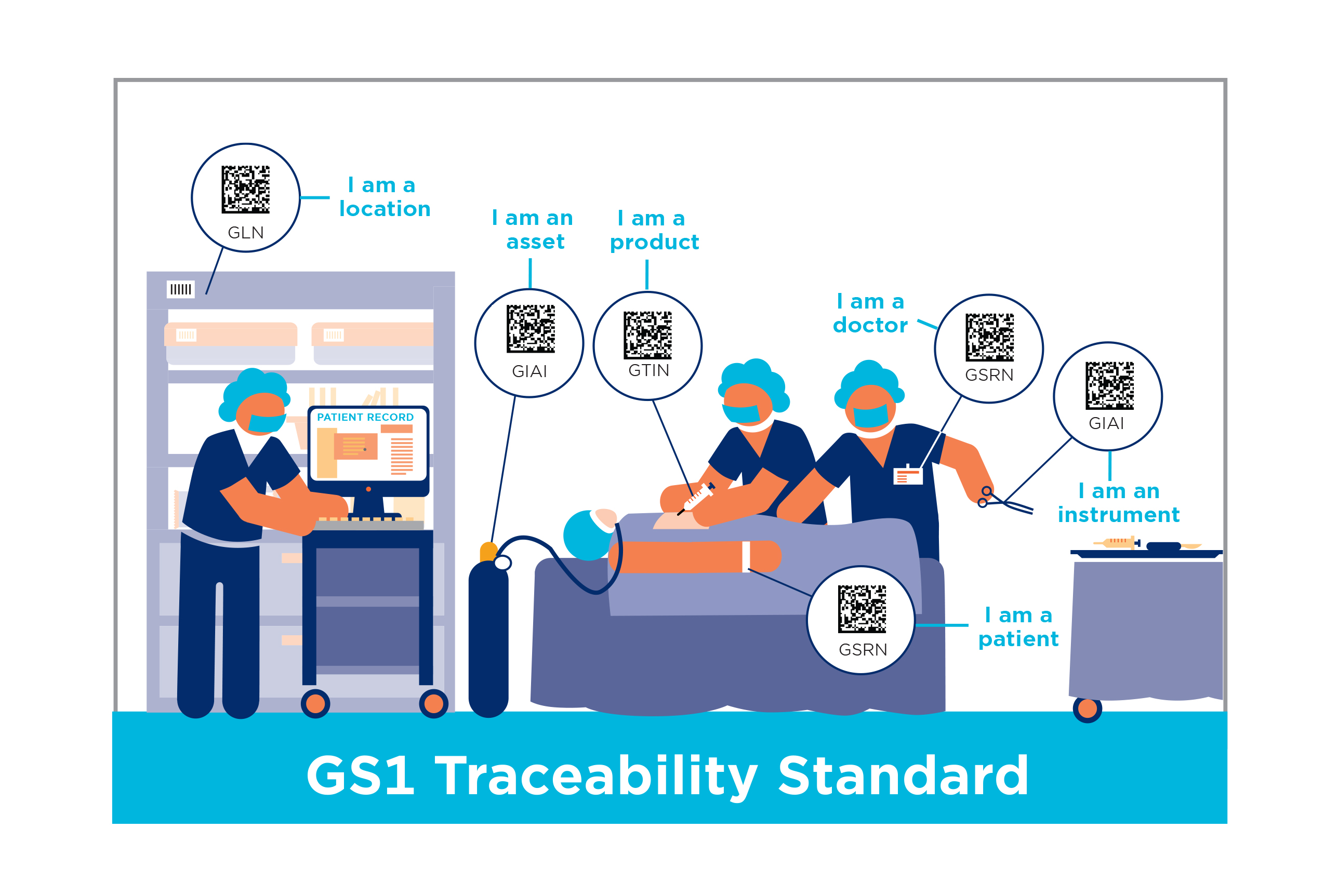
Applying GS1 Standards in Healthcare
Together, we're making it possible to follow drugs and medical devices from the manufacturer to the patient, improving patient care.
Hospitals are using GS1 Standards to ensure the reliability of data and optimise processes to reduce preventable errors, enhance Electronic Health Record management, support outcome-based medicine and comparative effectiveness, and provide transparency for better clinical and supply chain management.
Regulators around the world are looking for better traceability of product for both Pharmaceuticals and Medical Devices and the industry is leveraging GS1 Standards and the support of GS1 experts to prepare for and implement these requirements and to ensure regulatory compliance.

Healthcare Providers

Manufacturers & Distributors
Case Studies
Benefits on GS1 standards adoption in Healthcare
The opportunity to improve the performance of the Irish Healthcare service through use of standardised ICT, based on the GS1 System, is significant in terms of the potential for:
 Improved patient safety through the reduction of the risk of errors.
Improved patient safety through the reduction of the risk of errors.
 The provision of accurate master product information for medicines, medical devices and other products used to treat patients.
The provision of accurate master product information for medicines, medical devices and other products used to treat patients.
 Minimising waste - through better stock management including rotation to avoid goods exceeding their expiry dates.
Minimising waste - through better stock management including rotation to avoid goods exceeding their expiry dates.
 The elimination of enormous amounts of unnecessary paperwork, thereby releasing scarce people resources for other more added value tasks.
The elimination of enormous amounts of unnecessary paperwork, thereby releasing scarce people resources for other more added value tasks.
 Improved clinical data records through accurate and automatic pick-up of treatment information.
Improved clinical data records through accurate and automatic pick-up of treatment information.
 Ensuring the correct products are available when and where required.
Ensuring the correct products are available when and where required.
 The enhancement of traceability systems through information systems which automatically record batch/lot information.
The enhancement of traceability systems through information systems which automatically record batch/lot information.