- Home
- Industries
- Resources
- Case Studies
- Healthcare Case Studies
Loading...

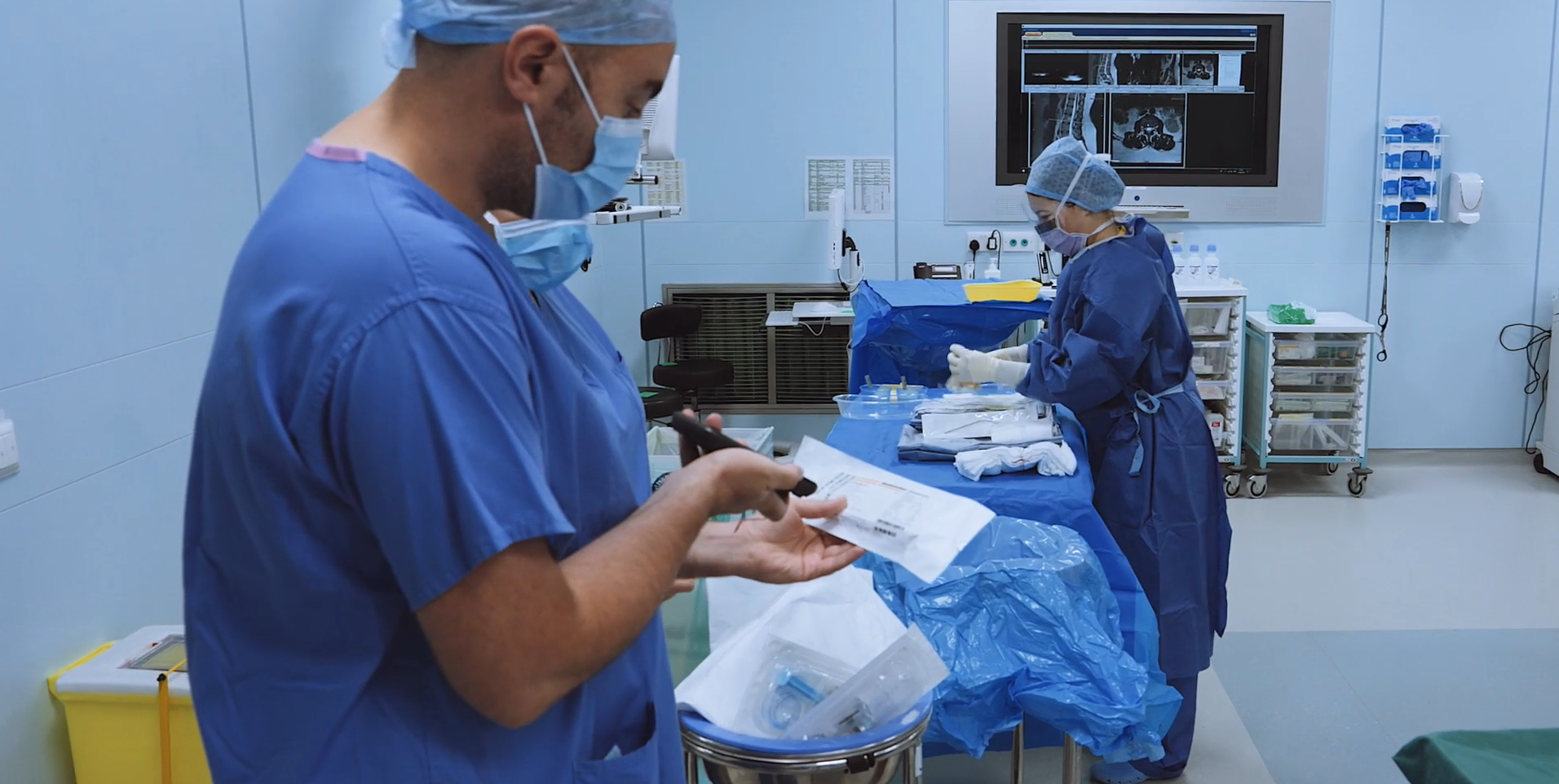
Scan4Safety: Giving time back to patient care at Dublin’s Tallaght University Hospital
The “Scan4Safety” project at TUH is part of a new digital strategy in the hospital with the objective of improved patient safety, with the added benefits of giving time back to patient care and increased operational efficiencies.

Traceability standards enable efficient rollout of COVID-19 vaccine.
As COVID-19 vaccines became available, Ireland’s Health Service Executive (HSE) needed an efficient and effective way of receiving, administering, tracking and reporting vaccinations across its more than 40 Centralised Vaccination Clinics (CVCs).

Best practice patient safety and traceability of infant feeds at CHI (Children’s Health Ireland) at Temple Street
The new electronic traceability system ensures complete, real-time visibility of each special feed across the hospital; in stores; in the feed preparation area; on the ward, right to the patient.

RFID-enabled transport of precious laboratory samples at St. James's Hospital
St. James’s Hospital in Dublin uses GS1 standards-based RFID technology to track the movement of laboratory samples throughout the hospital, a solution that is part of the hospital’s broader traceability strategy called PATH (Patient and Asset Tracking in Healthcare).

Haemophilia Treatment Traceability
Using an electronic patient record, unique product identification, barcode scanning and an innovative mobile phone app for patients St James's Hospital Dublin have been able to deliver a world class, integrated and comprehensive care solution for Haemophilia patients.

Patient Safety and Efficiency: St James's Hospital Whitepaper
Achieving World Class Patient Safety and Efficiency in Irish Healthcare - a white paper from St James's Hospital Dublin on its eProcurement practices utilising a central standardised product catalogue and electronic business messaging (EDI).

Scan for Safety /Scan for Surgery St. James’s Hospital – leading global innovation in healthcare
St. James’s Hospital has established the Scan for Surgery programme as well as introducing the automatic tracking of precious tissue samples. Both projects make use of GS1 standards-based technology to deliver improved patient safety and efficiency.